Radioactivity –what is it?
Different types of radiation
Radioactive substances have an unstable atomic nucleus that decays spontaneously, emitting energy-rich, or ionising, radiation in the process. This radiation is not detectable by human senses.
There are three main types of radiation:
- Alpha and beta radiation; these are produced as a result of the spontaneous alteration of an unstable nucleus into another nucleus.
- Gamma radiation; this is produced when an atomic nucleus emits excess energy.
Radioactivity decreases over time
Due to radioactive decay, radioactivity continuously decreases. This also applies to radiation from radioactive substances and thus also to the waste, which decreases over time until it reaches insignificant levels. For the protection of humans and the environment, this waste must be disposed of in a deep geological repository where safety barriers reliably enclose the waste.
Radioactive substances can be ingested through breathing or with foodstuffs and then decay inside the body (internal radiation). We can only protect ourselves from internal radiation by avoiding the ingestion of radionuclides.

Photo: © Fisfra | Dreamstime.com
Natural radioactivity
A small amount of natural radioactivity can be found everywhere: in rocks and soil, in building materials, in our food and in the air we breathe. Our bodies are thus continuously absorbing minuscule amounts of radioactive substances such as potassium-40 that is primarily incorporated into muscle tissue.
Natural radioactive substances were originally formed in exploding stars (supernovae) and then incorporated into our planet during its formation. Their decay makes a significant contribution to geothermal heat. To a small extent, natural radioactive substances also continue to form through interaction with cosmic radiation in the atmosphere.
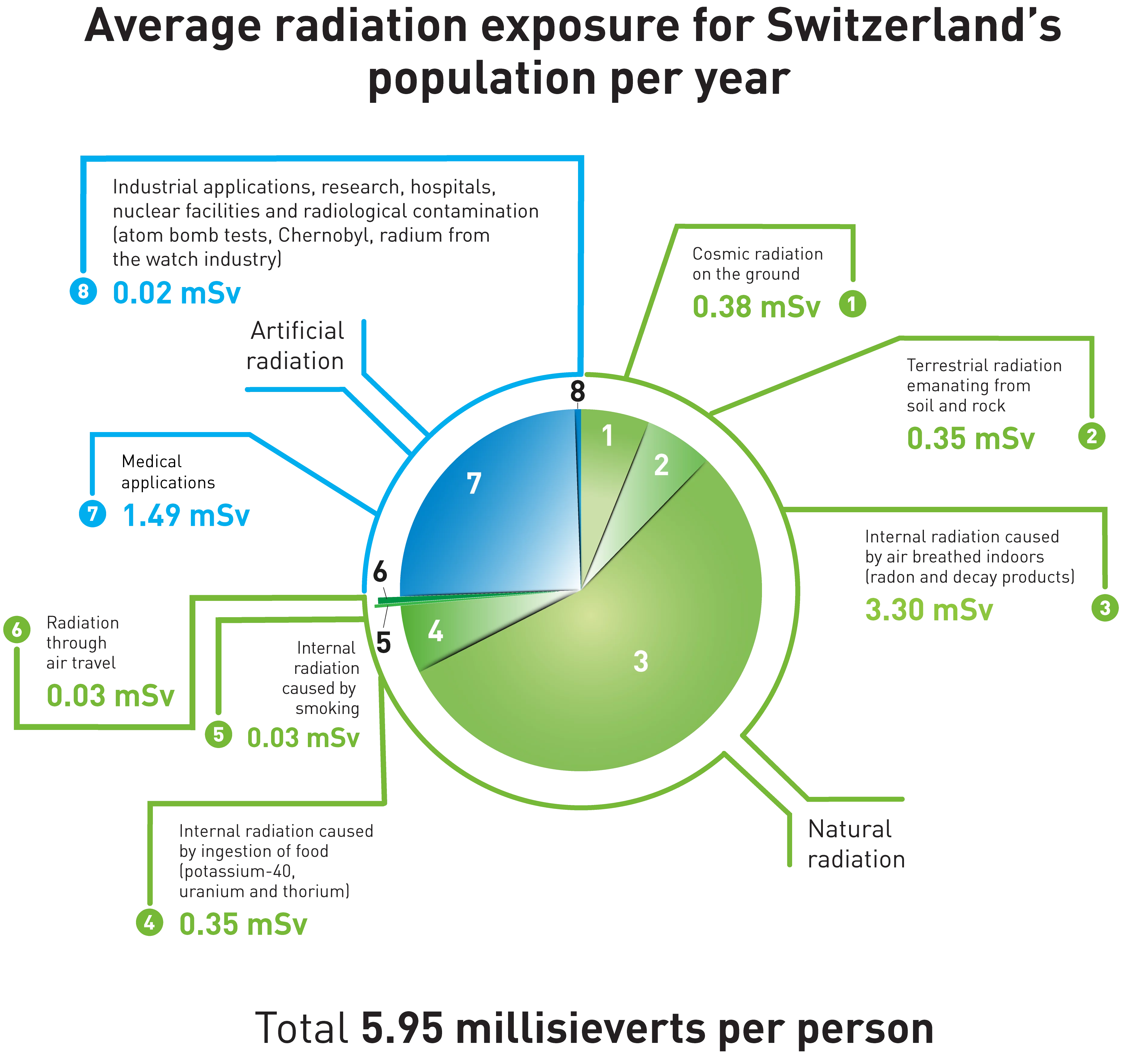
Natural radiation: radon by far the most common source
The average radiation exposure for a resident of Switzerland is around 5.8 millisieverts per year. Of this, the vast majority (4.3 millisieverts) is natural. This includes cosmic radiation from outer space, the intensity of which depends on the altitude at which we live. Some natural radiation comes from soil and rock; proportions vary by region. At 3.2 millisieverts, the highest dose from natural sources comes from gaseous radon in our homes (see Figure). This penetrates into the house from the underground, natural cellars or cracks, or is released by building materials in the house.
We are also exposed to artificial radiation. Higher doses are caused by medical applications and much lower doses due to industrial applications.
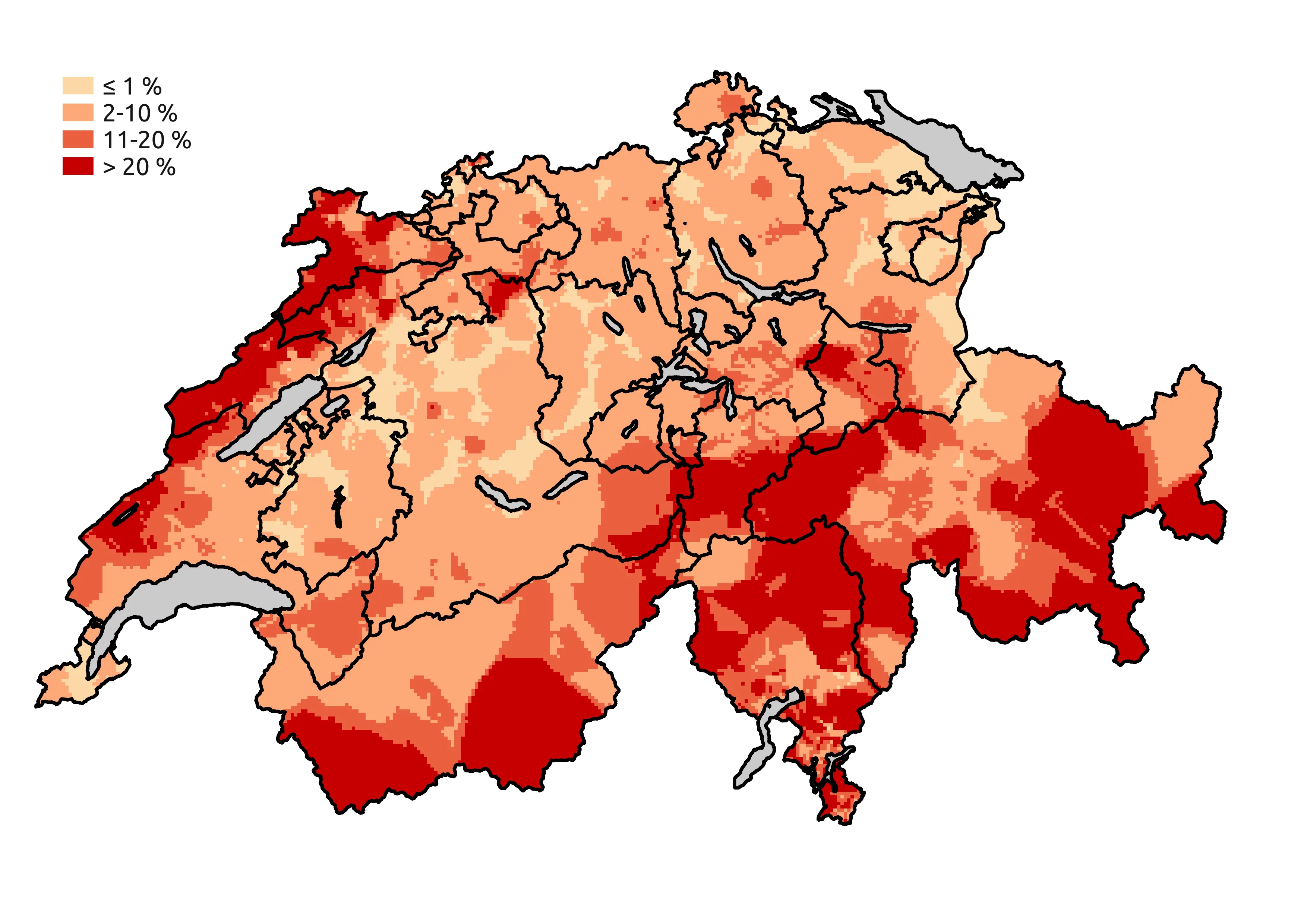
Avoiding radiation exposure
Alpha, beta and gamma rays are all forms of ionising radiation. They transfer so much energy to the atoms and molecules of the irradiated material that electrons are released from the atomic shell. This can cause chemical compounds to break up, resulting in damage to cells, tissue and organs.
The three types of radiation penetrate human tissue to different depths. Alpha rays have a short range and can only penetrate the top skin layer by fractions of a millimetre. Beta rays have a range of several millimetres and are therefore able to penetrate all skin layers. Energy-rich gamma rays fully penetrate the human body, spreading throughout the tissue and thereby slightly weakening the radiation.
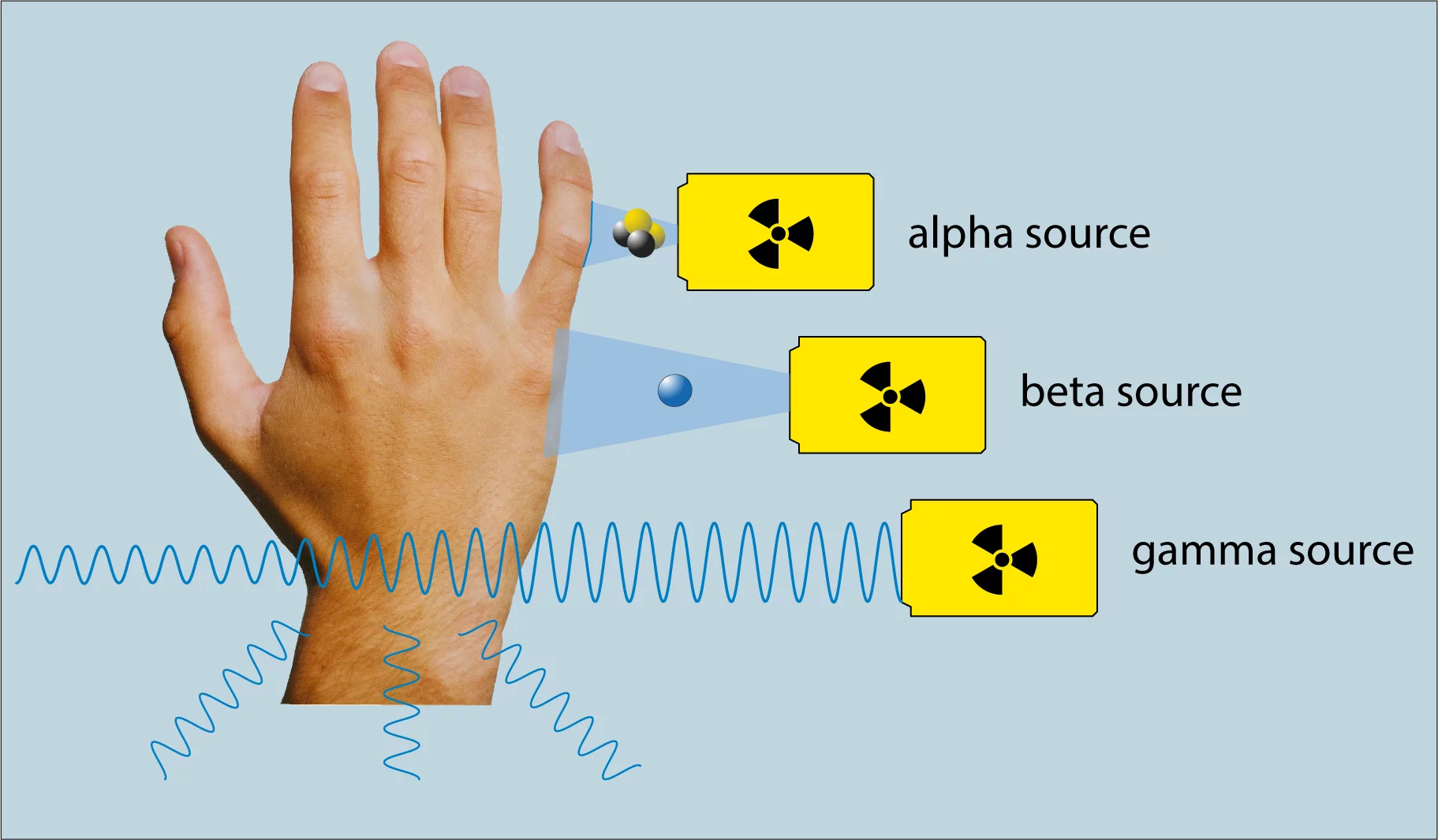
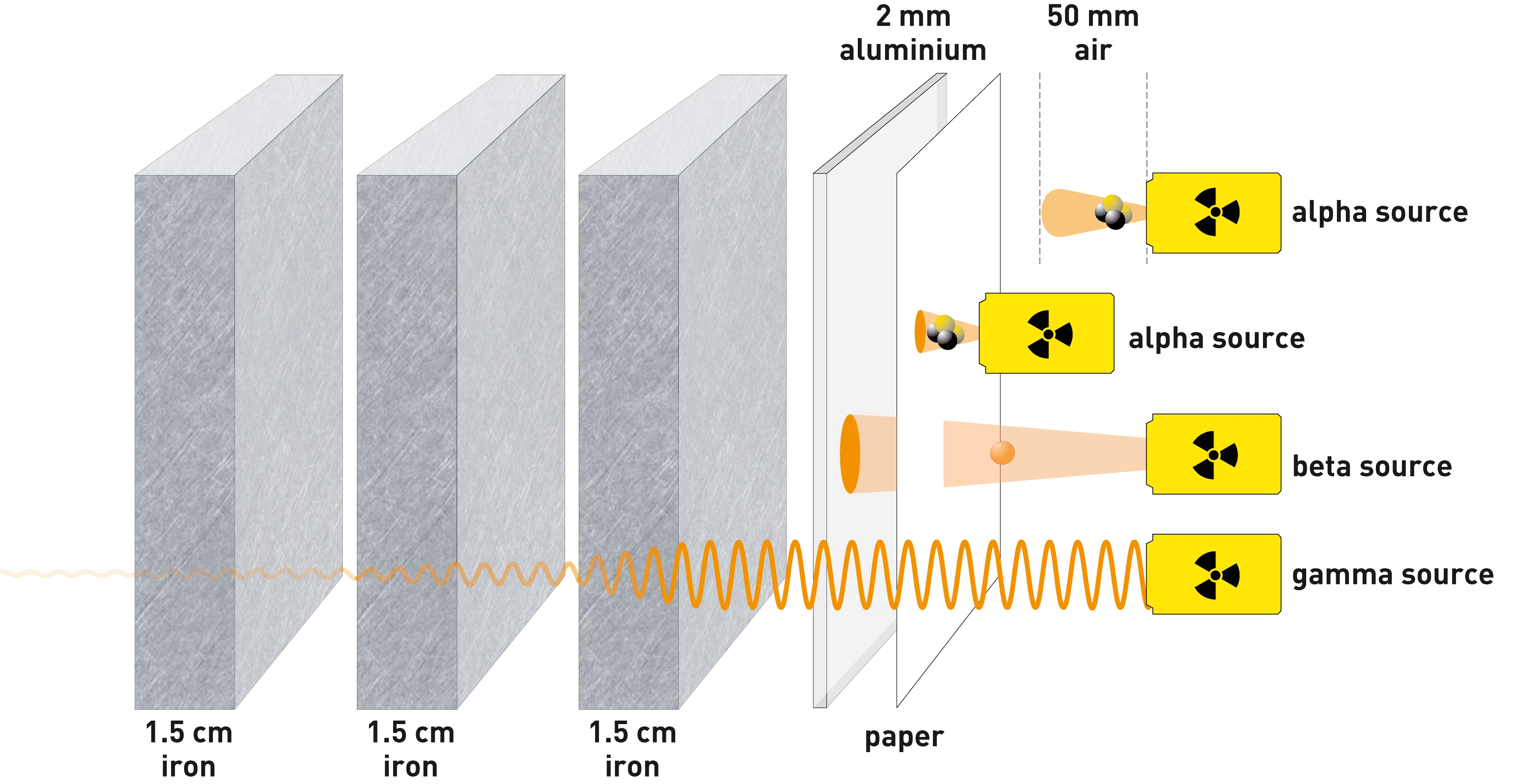
How can we protect ourselves from radiation?
Radiation from natural and artificial sources differs only in origin but not in its properties and effects. We can protect ourselves from a radiation source outside the body by using suitable shielding, keeping a safe distance and by limiting exposure time.
Alpha and beta rays can be completely shielded with relatively little effort. Energy-rich gamma rays can be weakened with a shield of several centimetres of iron or a few metres of rock or concrete.
Radioactivity is constantly monitored
The Swiss National Emergency Operations Centre (NEOC) operates its own network to measure radioactivity. Every ten minutes, 76 sensors distributed throughout Switzerland transmit current measurements. You can find the daily averages and progressions over time on the NEOC website.